Up until a decade ago, only a few antibodies had been identified as capable of significantly inhibiting HIV, and their activity was limited compared to antiretroviral drugs.
Antibodies are one of the more familiar components of the immune system, known for playing an important role in the body’s fight against many infections. HIV, like a number of other pathogens, has evolved mechanisms to fend off antibody attacks. In particular, HIV cloaks itself in a protective cloud of sugar molecules called glycans, which serves as a shield against antibody-mediated neutralization.
In most people with HIV, the immune system generates many antibodies against the virus—they are used as the basis for the HIV antibody test—but they are ineffective in blocking viral replication.
Over time, however, this area of research has been transformed by new technologies that greatly enhance the ability of scientists to discover and characterize rare antibodies with strong anti-HIV effects (see box: bNAb Discovery).
The upshot is that at the start of 2020, we have an ever-expanding family of antibodies capable of potently neutralizing a broad array of diverse HIV strains from around the world. They have been named broadly neutralizing antibodies (bNAbs). bNAbs are now being tested for HIV prevention, treatment, and even cure.
The current landscape of bNAb research
An antibody is a Y-shaped protein generated by B cells (a type of white blood cell that is part of the immune system). Their primary job is to bind to infectious agents, thereby neutralizing them and facilitating their elimination from the body. In some cases, antibodies also have the capacity to attach to cells infected with viruses or other pathogens, flagging the cell for destruction.
As the research progresses, bNAbs of increasing potency and breadth are being discovered. Potency refers to the amount of the antibody needed to inhibit HIV—the lower the amount of antibody required, the higher the potency. Breadth refers to how many different HIV variants can be inhibited. The virus is notorious for its ability to mutate and there are multiple different regional HIV subtypes (also called clades).
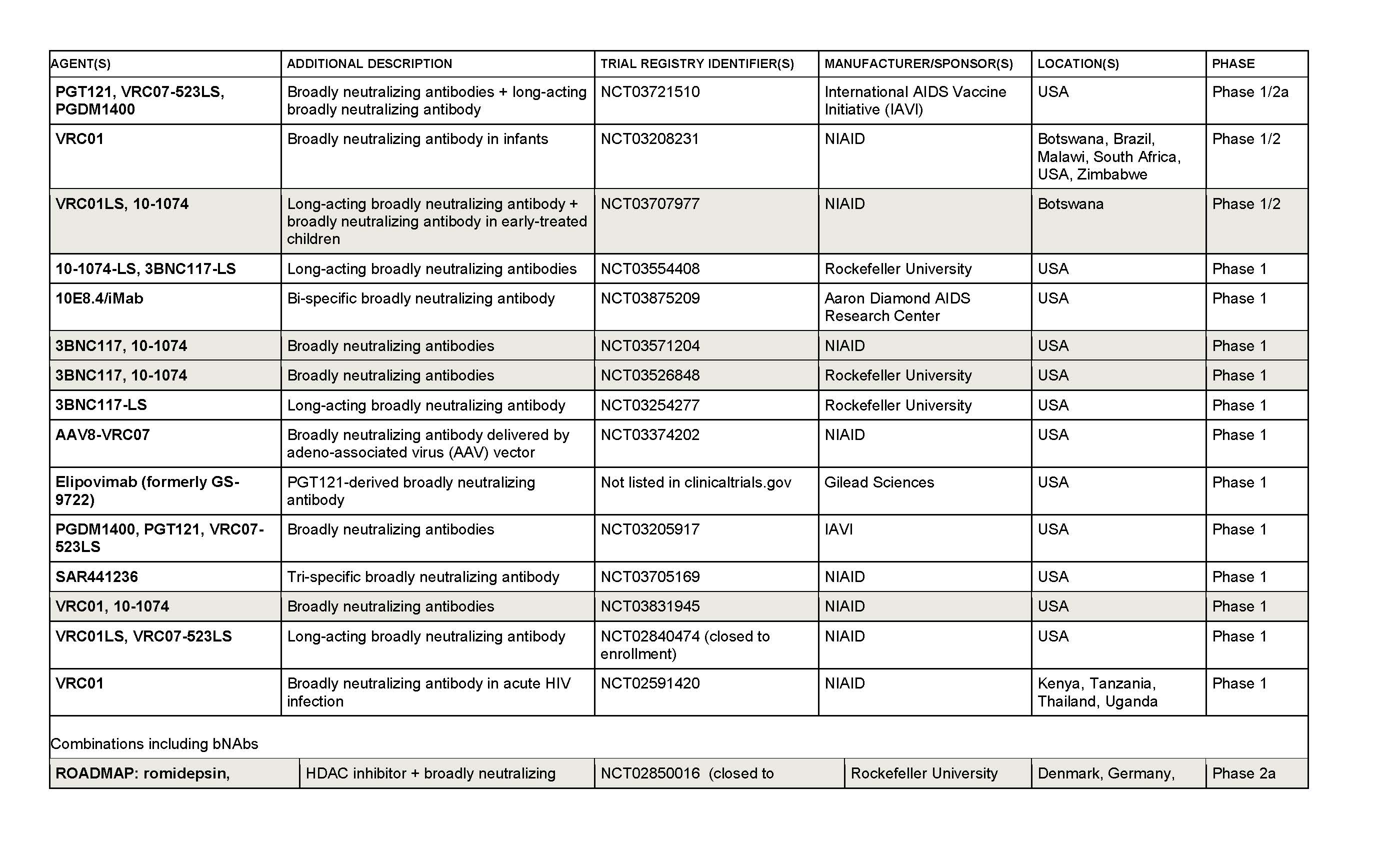
There are currently several threads to bNAb-related research in HIV (see also trial tables on following pages):
• The discovery, development and manufacture of bNAbs for delivery into the body, either singly or in combinations. This approach is being used to evaluate the ability of bNAbs to prevent, treat, or contribute to curing HIV when delivered via infusion, subcutaneous injection, or gene-based delivery systems.
• The development of preventive HIV vaccine candidates that aim to induce the production of bNAbs by the vaccine recipient’s B cells. This effort is at an early stage and is being guided by research into how bNAbs are generated by the immune system, as well as structural studies assessing how bNAbs interact with the parts of HIV that they target.
In experiments involving macaque monkeys and simian equivalents of HIV—either simian immunodeficiency virus (SIV) or SIV/HIV hybrids called SHIVs—bNAbs have demonstrated both protection against infection and an ability to significantly lower viral load.
The main Achilles heel is the development of resistance, similar to what has been seen with antiretrovirals when used alone. The future of bNAbs is therefore likely to involve combinations, particularly for therapeutic use.
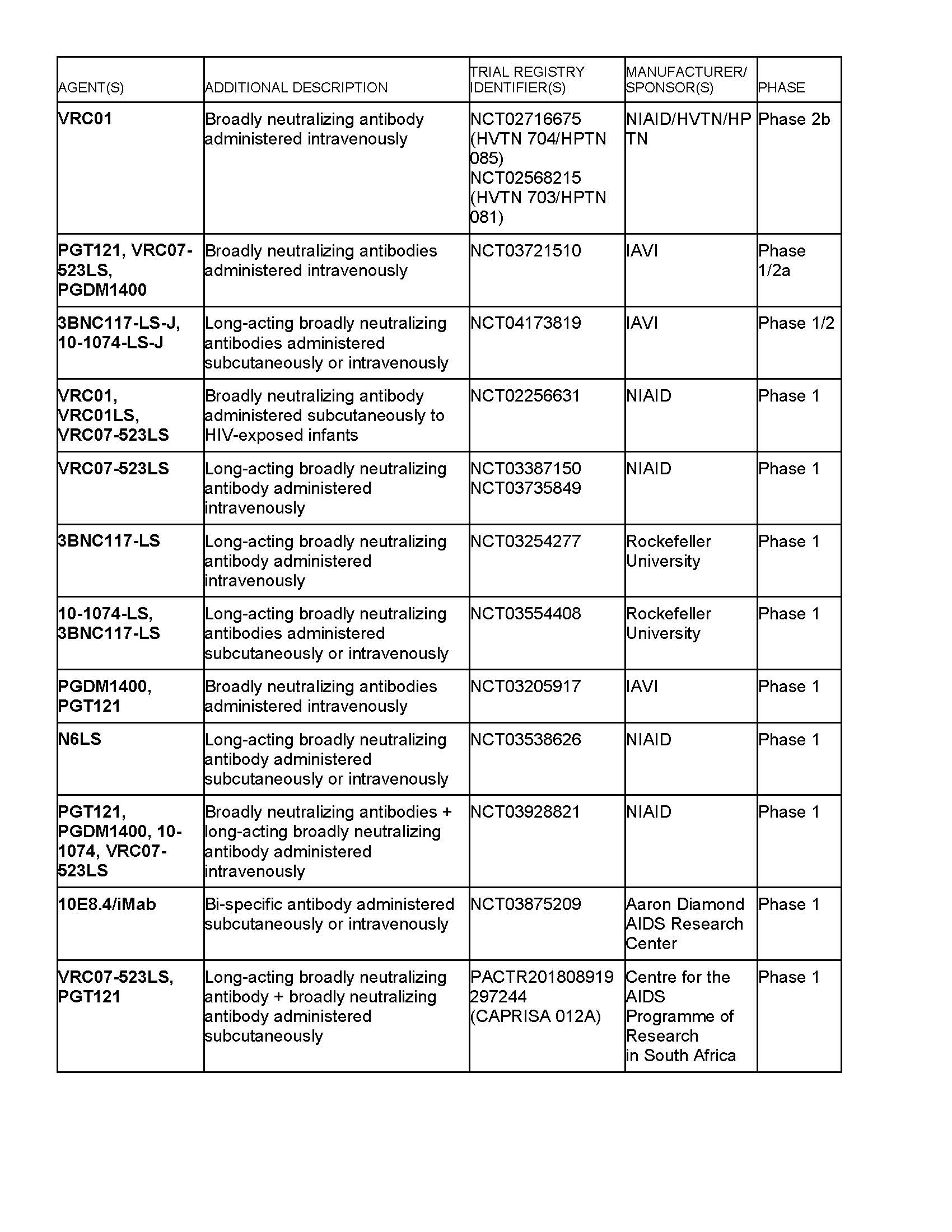
The largest ongoing human bNAb trials are the Antibody-Mediated Prevention (AMP) studies, which are evaluating whether bimonthly infusions of VRC01—one of the first bNAbs to be discovered—can prevent HIV acquisition in people at high risk of exposure. At the current time, individual bNAbs are mostly still known by the codenames assigned by their discoverers, making for a pipeline populated by candidates such as VRC01, 3BNC117, 10-1074, PGDM1400, N6 and PGT121. The shared feature of these bNAbs is that they are able to target sites of vulnerability on HIV’s outer envelope.
The AMP research is jointly sponsored by the HIV Vaccine Trials Network (HVTN) and the HIV Prevention Trials Network (HPTN), and the aim is to demonstrate “proof of concept” that bNAbs can offer significant protection against HIV.
The AMP studies comprise:
• HVTN 704/HPTN 085, which includes men and transgender individuals who have sex with men at sites in Brazil, Peru, Switzerland and the United States.
• HVTN 703/HPTN 081, which has recruited sexually active women at sites in Botswana, Kenya, Malawi, Mozambique, South Africa, Tanzania and Zimbabwe.
On June 21, 2019, HVTN announced that both trials had fully enrolled, with a total of 4,625 participants.
Even if efficacy is observed in the AMP studies, VRC01 probably won’t be advanced toward licensing for prescription because bNAbs with greater potency and breadth have since been discovered, and longer-acting formulations that offer less frequent dosing are also in the works (antibody names with “LS” added have been structurally modified to enhance their persistence in the body). The research aims to move the science forward by discovering how much bNAb needs to be present in the blood in order to achieve protection against HIV infection.
Several of these potentially improved bNAbs are in smaller, earlier phase trials both alone and in combinations. Delivery methods are typically either intravenous infusions or subcutaneous injections.
Researchers are also creating single antibodies capable of recognizing two or three different HIV targets, which are described as bi-specific or tri-specific—essentially a combination approach with one antibody. Both HIV-negative and HIV-positive individuals are being enrolled in studies due to the interest in exploring bNAbs in the preventive and therapeutic contexts.
Cure
For researchers working on attempting to develop curative interventions, there is interest in the potential for bNAbs to trigger destruction of HIV-infected cells and/or enhance immune responses against the virus.
The main mechanism for promoting killing of infected cells is antibody-mediated cellular cytotoxicity (ADCC). ADCC involves bNAbs binding to virus proteins displayed on the outside of a stricken cell and thereby attracting other immune system players such as natural killer cells to destroy the infected cell. Enhancement of immune responses against HIV could theoretically occur by bNAbs binding to HIV proteins and forming bundles called immune complexes, which can stimulate T cell immunity.
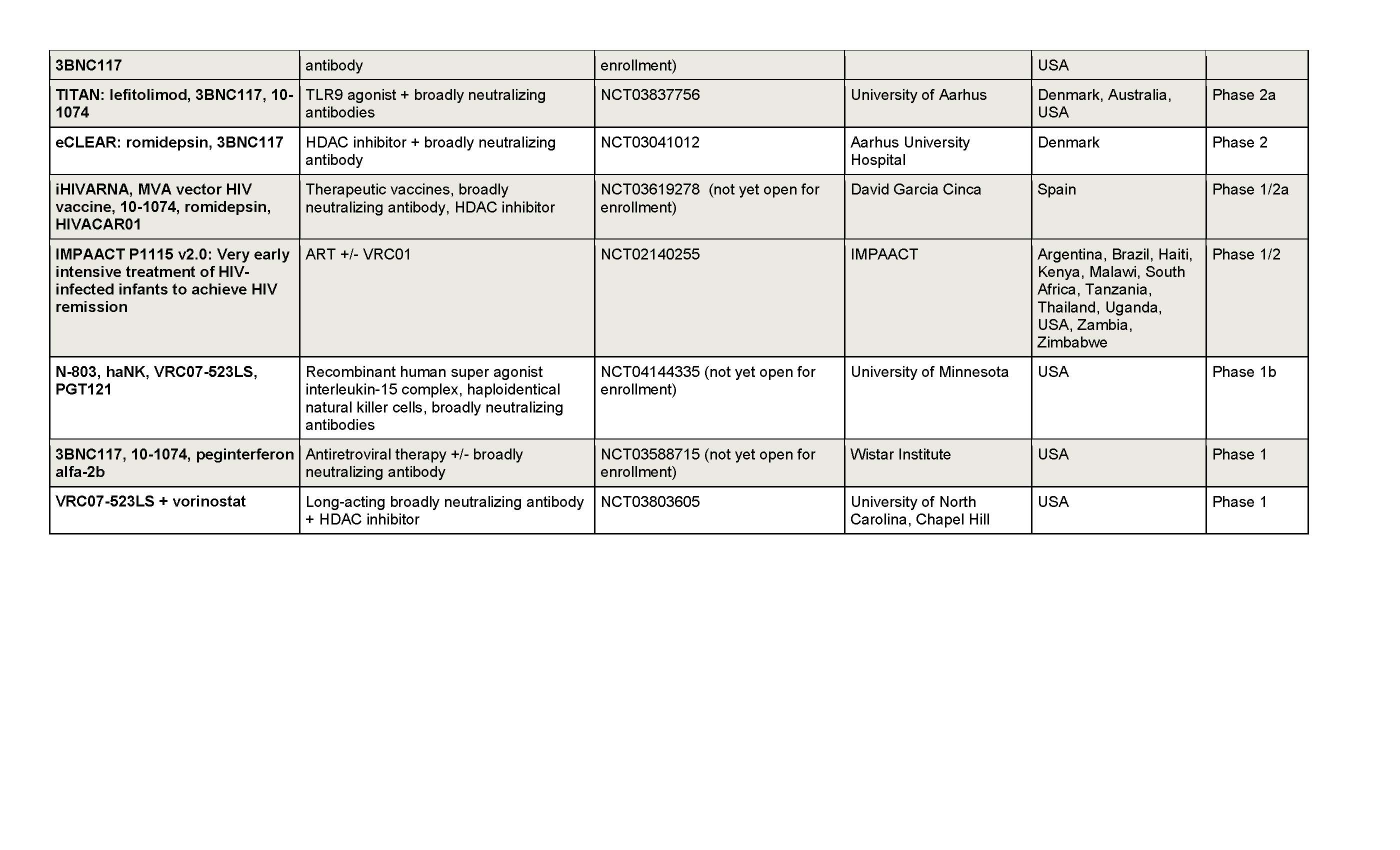
The enthusiasm for exploring these activities is reflected by the number of cure-related trials that are testing bNAbs or combinations of bNAbs with other interventions.
Several trials are attempting to target the reservoir of HIV that persists despite antiretroviral therapy (ART) by marrying bNAbs with strategies intended to awaken dormant or latent HIV, which would otherwise be invisible to the immune system. The hope is that these latency-reversing strategies will prompt virus-infected cells to reveal themselves by displaying HIV proteins on their surface, which can then be glommed onto by bNAbs, ultimately promoting destruction of the cell via ADCC.
HIV treatment interruption
The ability of bNAbs to maintain HIV viral load suppression during an ART interruption is another area of study. In some cases, the aim is to enhance the ability of the immune system to control HIV, but researchers are also assessing if ongoing intermittent bNAb administration could serve as a less burdensome means of maintaining viral load suppression compared to daily pill taking.
So far single bNAbs have shown limited activity after ART interruption and the development of resistance has been common, but in a small study with nine participants the dual combination of 3BNC117 and 10-1074 maintained viral load suppression for as long as dosing was continued. There were no cases of resistance developing to both bNAbs. Two of the participants continued to display suppressed viral loads until the end of the study period, 24 weeks after the last dose of the bNAbs, perhaps hinting at some enhancement of immune responses against HIV—this possibility is now being explored in additional trials.
Safety
No significant safety issues associated with bNAb administration have been documented to date among several thousand recipients, with one exception.
In a trial involving HIV-negative volunteers, subcutaneous delivery of the bNAb 10E8VLS prompted injection site reactions, including quite severe skin reddening and irritation (erythema), along with fever and malaise. Researchers suspect that the adverse events may be linked to the way this specific bNAb interacts with lipid molecules on the surface of cells, and this is being investigated (the trial has been suspended).
Production challenges
In the ideal scenario in which bNAbs are able to demonstrate sufficient efficacy to justify licensure—whether in the preventive or therapeutic realms—the next challenge will be to ensure that they are affordable and globally accessible.
Manufacturing antibodies is significantly more complex than the production of most pharmaceutical drugs. The process is expensive, involving the use of cell lines to churn out the antibodies in vast steel tank bioreactors. Estimates suggest production costs of around $100 per gram of bNAb, and this would need to be drastically reduced to around $3 per gram to ensure equitable access.
Efforts are underway to address the problem. IAVI is collaborating with the Serum Institute of India, an established vaccine manufacturer, to explore strategies for reducing production costs. In parallel, researchers are continuing to work on increasing the potency and persistence of bNAbs in order to reduce the amount that would need to be administered.
At least two large pharmaceutical companies are in the bNAb mix. Gilead Sciences licensed the rights to the bNAb PGT121 and are now testing a modified version, which has the distinction of being the first to be given a name: elipovimab (formerly GS-9722). More recently, ViiV Healthcare announced that they have acquired the rights to commercially develop the bNAb N6LS for HIV treatment and prevention.
The future for bNAbs
For a long time, it was thought that HIV might essentially be invulnerable to antibodies. The technological revolution of the last decade has overturned that pessimistic assumption, leading to the alphabetical and numerical soup of bNAbs now in clinical trials. Progress to date suggests that even more potent and broadly active bNAbs are likely to be on the horizon.
Until additional results become available, the role that bNAbs will play in the future of HIV prevention and treatment is uncertain. But there are reasons to be optimistic about their potential to add new, relatively user-friendly options to the current array of interventions.
bNAb Discovery
The technologies that have facilitated the discovery of bNAbs allow researchers to sample huge numbers of B cells from an individual, and then analyze the anti-HIV effects of the antibodies being made by each individual B cell.
Sampling B cells from people with HIV has led to the discovery that some people generate rare antibodies with the capacity to strongly inhibit the virus. In most cases the antibodies are not of benefit to the sample donor, because they’re present in only small amounts and are essentially overwhelmed by the virus.
Researchers can pinpoint the genetic code of the B cell that acts as a blueprint for producing the bNAb, and then use that code as a means to manufacture the antibody in large quantities.
AAV-based bNAb delivery
One ongoing trial is assessing whether it might be possible to avoid intravenous or subcutaneous injection of bNAbs by using a delivery method drawn from gene therapy. The genetic code for the bNAb VRC07 is inserted into a harmless adeno-associated virus (AAV), which is administered by a single injection. The idea is for the AAV to persist in the body and act as a factory that produces a constant supply of the bNAb. A similar strategy has been used with some success to deliver factor IX to hemophiliacs. However, an initial study in HIV-negative volunteers sponsored by the International AIDS Vaccine Initiative (IAVI) found that the body generated antibodies against the bNAb encoded by the AAV, preventing the achievement of detectable bNAb levels—this problem will need to be overcome in order for the approach to be successful.
Richard Jefferys began working in the field of HIV/AIDS in 1993 as a volunteer at the nonprofit AIDS Treatment Data Network in New York City. He joined Treatment Action Group (TAG) in 2001, and directs TAG’s Basic Science, Vaccines, and Cure Project.
Go to clinicaltrials.gov and enter the trial registry number to learn how to take part in the study
SHADED ENTRIES INDICATE INCLUSION OF AN ANALYTICAL TREATMENT INTERRUPTION (ATI).
See “Briefly” for an update on the risk of HIV transmission related to ATIs.
Go to clinicaltrials.gov and enter the trial registry number to learn how to take part in the study