
With an estimated population of 652,405 (as of 2013), Seattle is the largest city in the State of Washington. The city’s skyline is dominated by the 605-foot tall Space Needle, built for the 1962 World’s Fair. Looming in the distance is Mount Rainer, 54 miles southeast of Seattle, at a height of 14,411 feet.
The Conference on Retroviruses and Opportunistic Infections, or CROI, has long been an important gathering for clinical scientists, doctors, and epidemiologists to discuss the latest research and study results for the prevention and treatment of HIV/AIDS and opportunistic infections. Here are some of the most important news items from CROI 2015, held from February 23–26 in Seattle, Washington.
BY ENID VÁZQUEZ
HIV can be prevented with the use of four pills taken around the time of sex. CROI 2015 brought this and a lot of other good news on using the HIV medication Truvada to prevent infection with the virus in HIV-negative people (an intervention called PrEP, for pre-exposure prophylaxis).
Meanwhile, researchers reported zero linked infections in nearly 6,000 acts of condomless anal sex in 152 male serodiscordant couples when the HIV-positive partner had undetectable viral load on therapy, a strategy called Treatment as Prevention, or TasP (Grulich, 1019LB).
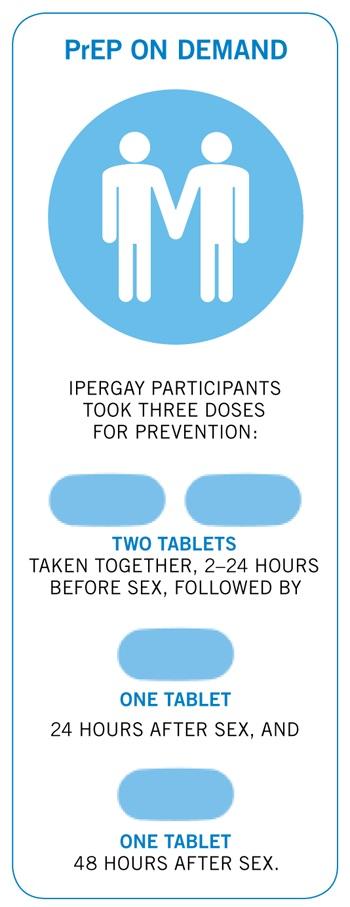
On demand PrEP
Doctors from Montreal and Paris teamed up to study Truvada PrEP around the time of sexual risk, and found an 86% decrease in risk of HIV with the strategy.
The IPERGAY study design was based on one in macaque monkeys showing “you almost fully protect” them with Truvada before and after exposure to virus, said presenter Jean-Michel Molina, MD, of the HIV research collaborative ANRS in Paris.
Participants used three doses totalling four tablets for prevention. Anyone having sexual intercourse more than once in a week was advised to continue taking one pill a day, followed by the two doses after the last encounter. Altogether, most people used sixteen tablets a month, for an average of one sex act a week.
Then again, participants did not always use PrEP. “Overall, 28% of these participants did not use PrEP during the last intercourse, so patients were able to select the type of sexual [act] including the type of partner with whom they would want to use PrEP,” Molina said.
Wait for confirmation of the results before trying this at home.
Among its conclusions, the research group reported that on demand PrEP is an “attractive alternative to daily PrEP [as FDA approved] in high-risk MSM [men who have sex with men] who do not use condoms consistently.”
The group expected to show at least a 50% decrease in HIV incidence and the 86% reduction they saw surprised them. Due to the success of the Truvada group in staying HIV free, the placebo arm was discontinued early and everyone in that group offered the opportunity to take the drug.
Real-world PrEP
British researchers decided to check on the PrEP potential in the real world following a 44% reduction in HIV seen in the landmark iPrEx study.
Presenter Sheena McCormack, MD, noted that effectiveness might be less in the real world because iPrEx participants were seen monthly and had strong resources for adherence support, and because they received behavioral interventions and were constantly reminded that PrEP may not work.
Would people take greater risks knowing that Truvada PrEP does work to prevent HIV?
As with IPERGAY, taking Truvada succeeded in achieving an 86% reduction in new HIV infections. Participants were recruited from sexual health clinics in London.
These men also needed to be at high risk of infection. They reported condomless anal sex in the past 90 days and were likely to have condomless sex again.
Also as with IPERGAY, the Truvada PrEP group did so well at preventing HIV that the data monitoring board recommended giving the drug to all participants in the study. And again as in IPERGAY, the research team was surprised at how many infections they saw in the group not given Truvada. It was three times the number of HIV infections they expected to see.
The group concluded, among other things, that, “Our concerns about PrEP being less effective in the real world were unfounded.”
“What is absolutely fantastic,” said McCormack, “is that given [the successful] results for PrEP, people who need it really came forward.”
¡Adios, HIV!
Not mincing their words, the Partners Demonstration Project researchers called the results of their study with Truvada “the near elimination of HIV.”
While high levels of adherence to either PrEP or ART (HIV therapy) can reduce the risk of infection by more than 90%, said presenter Jared M. Baeten, MD, PhD, of the University of Washington, there are issues to consider. Delays in starting HIV therapy are common, and transmission risk may continue for months after starting treatment until a person’s virus is undetectable. Some studies have reported that not everyone offered HIV therapy wishes to take it. As for PrEP, not all at-risk individuals use it when it is offered.
This study looked at PrEP as a prevention bridge in the HIV-negative partners of serodiscordant couples in four African countries, hoping to keep them uninfected until the positive partners get their virus down to undetectable through therapy. It found that PrEP reduced infections by 96% (from the expected rate of more than 5% a year down to a half-percent).
“Our results also speak to the potential for HIV protection from integrated PrEP and ART in other high-risk populations, we would argue, including MSM and women at risk,” said Baeten.
San Francisco
Using a mathematical model, leading medical providers from San Francisco showed that the city can reduce its number of new HIV infections by 70%.
Presenter Robert M. Grant, MD, of iPrEx study fame, said that although PrEP use was “negligible” when it became available in 2011, it increased over time. In turn, increasing PrEP use correlates with decreasing numbers of new HIV infections, he said.
New infections went down from 420 in 2012 to 359 in 2013, and so far for 2014, have gone down to 320 (some of the cases need to be confirmed, so the number may be even lower).
As with the Partners Demonstration Project, the San Francisco group expects HIV treatment and PrEP to bolster each other’s effectiveness for prevention, and as with the London group, it noted that people at highest risk for HIV had the greatest uptake of PrEP.
“Remarkably, the uptake of PrEP is strongly and positively correlated with reported HIV risk, such that 63% of men reporting condomless anal sex in the last six months reported having used PrEP,” said Grant, who is now Chief Medical Officer for the San Francisco AIDS Foundation. “People with lower levels of risk have much less uptake.”
WATCH WEBCASTS OF THESE PREP FINDINGS AT THE CONFERENCE WEBSITE, CROICONFERENCE.ORG.
Body composition
The early days of highly active medication for HIV were known for resulting in undesirable body composition changes. Although the causes of these changes are not completely understood to this day, the AIDS Clinical Trials Group (ACTG) presented data showing that the virus itself may not only contribute to increased fat, but do so more than the medications.
Presenter Grace A. McComsey, MD, of Case Western Reserve University, said that lipoatrophy (fat loss in the face and limbs) wasn’t seen with newer medications, but fat gain was.
“A 30% gain in fat after two years? That’s really bad,” she said in response to a question from the audience. “It’s a relatively short duration of treatment.”
The effects were similar no matter what regimen people took.
It was the strong correlation with the virus that came as an even greater surprise.
“I think it’s amazing that regardless of the regimen you looked at, the fat gains were so much more if you start with high viral load [over 100,000],” McComsey continued. “Then, if you adjust for inflammation markers, viral load remains by itself associated with higher gains. The research, especially on central fat, needs to continue and needs to be geared towards inflammation and other markers, rather than just the drugs.”
The metabolic sub-study of A5257 looked at fat loss and increases along with measurements of inflammation in 328 individuals who were using regimens containing Isentress (raltegravir, an integrase inhibitor) or Prezista (darunavir) or Reyataz (atazanavir), both protease inhibitors (PIs).
Although the protease inhibitor class of drugs was thought to be responsible for fat gain back in the day, this study found no difference in fat gain between Isentress and the two PIs.
Lipitor
In a small study with 40 individuals living with HIV, half were put on the statin medication Lipitor (atorvastatin).
“As compared to placebo, statin therapy reduces noncalcified plaque volume and high risk plaque features in HIV-infected patients with subclinical coronary atherosclerosis,” researchers reported.
Plaque consists of fatty deposits and other types of cells that build up in arteries. This can cause the progressive disease atherosclerosis.
The group from Harvard Medical School wrote that, “Further studies should assess whether reduction in high risk coronary artery disease may translate into effective prevention of cardiovascular events in this at-risk population of HIV patients.”
The team noted that while drugs known as statins can reduce the risk of cardiovascular disease events and deter the progression of atherosclerosis, “no studies have yet assessed the ability of statin treatment to achieve regression of coronary atherosclerosis in HIV-infected patients, a population demonstrated to have elevated risk of myocardial infarction [heart attack].”
Kaletra vs. Viramune for children
The IMPAACT P1060 study had earlier reported short-term superiority of Kaletra over Viramune (nevirapine or NVP) for infants through 24 weeks of life, regardless of any exposure to Viramune for HIV prevention around the time of birth. “In contrast,” the research group wrote, “those on NVP had marginally superior improvements in CD4% and growth (weight and height z-scores).”
Now the group reports that in longer term follow-up, “Early modest gains in CD4% and growth associated with NVP were no longer statistically significant beyond one year after ART initiation.”
“These findings further support the current WHO recommendation for LVP/r [Kaletra]-based ART as first-line therapy for HIV-infected children aged three or younger,” they wrote.
Short-cycle therapy
Teenagers put on five days of therapy followed by weekends off continued to have the same treatment success as those continuing with daily HIV medication. At one year, the results were similar for viral load, safety, virus drug resistance, and signs of inflammation.
The BREATHER Study included 199 young people from 11 countries in Europe, Thailand, Uganda, Argentina, and the U.S. Before entering the study, all of them had undetectable viral loads under 50 for at least one year with efavirenz (Sustiva, called Stocrin in the UK, Europe, and elsewhere) and two nucleoside drugs (such as Viread and Emtriva).
Karina Butler, of Our Lady’s Children’s Hospital in Dublin, said the teenagers preferred taking a couple of days away from treatment on the weekends. They were happy not to have to take their HIV medications during a sleepover, for example.
There is also a cost savings with the fewer pills. For now, the abbreviated regimen is not approved for use.
BY JEFF BERRY
Tenofovir disoproxil fumarate (TDF), a component of Truvada and included in most HIV regimens used to treat HIV, is associated with clinically significant renal and bone toxicity. TDF is a prodrug of the active drug tenofovir diphosphate (DP). TDF is converted to the active tenofovir in the blood plasma, then makes its way into the cells.
A new “version” of TDF, tenofovir alafenamide fumarate (TAF), is also a prodrug of tenofovir DP but is metabolized largely within the cell, making levels in the blood plasma much lower (90% lower in studies), resulting in reduced side effects and increased concentrations of the drug within cells. So there has been a lot of interest in seeing this drug move forward.
David Wohl, MD, presented Phase 3 48-week data in a planned analysis from two identically designed, international, randomized, double-blind active-controlled studies (104 and 111) in 1,733 individuals. The two studies compared a once-daily single-tablet regimen (STR) of elvitegravir 150 mg/cobicistat 150 mg/emtricitabine 200 mg/TAF 10 mg (E/C/F/TAF) to the currently available Stribild, an STR which contains TDF (E/C/F/TDF). Both studies were in treatment-naïve individuals who had a viral load equal to or greater than 1,000 copies/mL and an eGFR (glomular filtration rate, a marker of kidney function) of at least 50 (lower than the current Stribild threshold of 70).
The median age was 34; 50% of the participants were female, 25% black or African descent, and 19% Latino. The average viral load of study participants at baseline (when they started) was 4.58 log10, with 23% having a baseline viral load over 100,000, and 5% over 400,000 c/mL. The median CD4 count was 400.
TAF and TDF achieved comparable levels of viral suppression in patients with viral load above and below 100,000. Similarly for CD4 count, those with CD4 count less than 200 saw no difference in rates of viral suppression. Results were similar across all groups when broken down by age, race, and sex. There was a “robust” CD4 count increase during the trial, and at week 48 there was a greater increase of 30 CD4+ T-cells in the TAF group versus the TDF group (211 vs. 181), which was statistically significant. The rate of virologic failure was 4% in both groups, while emergent resistance was less than one percent.
Safety was “well-balanced” between both arms in the two studies. Any serious adverse event (AE) was less than 10% in both arms, with less than 1% in the TAF group and 1.5% in the TDF group experiencing a treatment-related discontinuation. There were varying reasons given for treatment discontinuation, but as Wohl stated, “importantly in the TAF arm there was no new signal or anything to differentiate between the two arms, and no discontinuation due to renal or bone toxicity in the TAF arm.”
Overall 92% treated with E/C/F/TAF achieved virologic suppression through week 48 in this combined analysis of two “very well-powered” clinical trials, demonstrating non-inferiority to E/C/F/TDF.
In a separate presentation (143LB) at the conference Paul Sax, MD, presented a subset of data on renal and bone safety from the two studies. The decrease in spine and hip bone mineral density (BMD) was significantly less in the TAF arm compared to the TDF arm, and there was less of a GFR decline in TAF vs. TDF. Increases in lipids were greater in the TAF arm, including HDL (the “good” cholesterol).
A related poster at the conference showed that patients with mild to moderate renal impairment (eGFR 30-69 mL/min) who switch to E/C/F/TAF saw bone mineral density and markers of kidney function improve through 48 weeks (Pozniak, Poster #795).
Maturation inhibitor BMS-955176
Novel antiretrovirals that target different steps of the virus life cycle, have unique resistance profiles, and display long-term tolerability with manageable drug-drug interactions are important, especially for treatment-experienced patients who may have limited options due to treatment emergent or transmitted resistance, and/or regimen complexity.
Maturation in the viral life-cycle process is especially important, because it’s the point at which the virus changes structure and becomes infectious. Maturation inhibitors (MIs) bind to the Gag polyprotein of the budding virus and block a key protease cleavage event (past the point where protease inhibitors, or PIs, work), thereby blocking maturation.
176 is a second-generation MI in development by Bristol-Myers Squibb (BMS) with no significant safety issues identified to date. This was a Phase 2a dose-ranging study in 60 individuals who were either treatment naïve (92%) or experienced (PI and MI naïve), with a viral load equal to or greater than 5,000 copies, and CD4 count greater than 200 who received doses of 5, 10, 20, 40, 80, or 120 mg or placebo once daily (QD) for 10 days. The study was conducted in Germany in mostly white males, median (half above, half below) age 37, median viral load 4 logs, and median CD4 512. Median change drop in HIV-1 RNA from baseline to day 11 was 1.4 log10.
The greatest response was with the 40 mg dose. In its conclusion, the study team reported that, “Unlike 1st-generation MIs, in this proof-of-concept study BMS-955176 showed similar antiviral activity in subjects with wild-type HIV-1 or HIV-1 with Gag polymorphisms. BMS-955176 was generally well tolerated at all doses.”
TLR7 GS-9620
As discussed in the cure article in this issue, research is looking for ways to eliminate the HIV reservoir, where the virus lays sleeping in the body (called “latency”). One method seeks to “kick” the virus out of its hiding spot so it can be targeted and destroyed by HIV treatment. There was exciting data about a new drug in development from a class called TLR7 agonist.
Increased HIV levels in the test tube were seen when GS-9620, being developed by Gilead Sciences, was added to blood cells donated by four HIV-positive individuals. Another small study showed that the drug worked well in monkeys and is going on into human research.
A third research team noted that, “Pharmacologic activation of latent HIV reservoirs is considered to be a key part of the strategy towards eradicating HIV-1 infection,” and reported that GS-9620 was successful in activating the virus in the blood cells of HIV-positive patients (specifically, peripheral blood mononuclear cells, or PBMCs). The team said that together with the monkey data, the results support further research with HIV-positive people on therapy with undetectable virus “for possible activation and reduction of the viral reservoir.” This study has been initiated.
According to a report from Project Inform, “Toll-like receptors [TLRs] are receptors on immune system cells that, among other things, can result in the production of proteins that regulate the immune response to viruses, bacteria, and other pathogens. … TLR7 has been implicated in autoimmune responses (where the body attacks itself), so it is something of a surprise that the TLR7 agonist being studied by Gilead appears to be safe to use. [GS-9620] is already in Phase 2 studies as a treatment for hepatitis B, and experience with the drug led scientists at Gilead to believe that it might be effective in a kick-and-kill model aimed at reversing HIV latency.”
Abacavir and heart attacks
New data was presented on the possible link between heart attacks and the HIV medication abacavir (brand name Ziagen, also found in Epzicom and Triumeq). Once again, research found greater risk from traditional factors for myocardial infarction (MI, or heart attack).
Looking at 301 heart attacks among 16,733 individuals with HIV here in the U.S., the NA-ACCORD study reported that, “We found an increased risk for MI with recent [within the past six months] ABC [abacavir] use that diminished in magnitude and statistical significance after adjusting for traditional and HIV-associated MI risk factors, many of which were significantly more prevalent in ABC users. Further analyses are underway to account for potential time-dependent confounding of risks for MI.”
The results showed a 34% higher risk of MI (after adjustments), but this was not a statistically significant difference.
People who went on abacavir had greater risks, in part, because their medical condition did not allow them to go on the drug’s main competitor, tenofovir DF (for example, the presence of kidney disease).
BY ANDREW REYNOLDS
PROJECT INFORM
In recent years, other infectious diseases besides HIV have been featured prominently at CROI, most especially hepatitis C (HCV). CROI 2015 was no exception and with more than 100 abstracts, HCV research was well represented. This brief review provides information on the latest data from a selection of posters and presentations, with particular emphasis on
HIV/HCV co-infected persons.
Curing HCV: ION-4 and ALLY-2
Today, co-infected patients are more likely to experience the same cure rates as those infected only with HCV.
Prior to the direct acting antiviral (DAA) era, treating HCV in HIV/HCV co-infected persons was very challenging: Pegylated interferon and ribavirin were the standard of care and the combination of length of treatment, severe side effects, and low success rates stood as significant barriers for patients and their medical providers.
In the DAA era, we are not only seeing high sustained virologic response (SVR, or virologic cure) rates in HCV mono-infected people, but we are also seeing nearly the same rates in co-infected people. Indeed, the AASLD/IDSA/IAS HCV treatment guidelines recommend that HIV/HCV co-infected persons should be treated and re-treated the same as those without HIV (once drug-drug interactions have been addressed). Several presentations at CROI provide further support for both the FDA-approved and off-label use (when there is no FDA approval but evidence of potential benefit exists) of DAAs in co-infected patients.
The fixed-dose combination of ledipasvir/sofosbuvir, brand name Harvoni, was approved by the FDA in October 2014 for the treatment of HCV in genotype 1 in mono-infected persons, but not for HIV/HCV co-infection. Although we have seen some very compelling evidence that this treatment is effective in co-infected persons—an NIH study comprising 50 non-cirrhotic HIV/HCV co-infected patients had an SVR rate of 98% (49 of 50)—the numbers were too small to draw any conclusions regarding its efficacy in larger groups.
Results from the ION-4 demonstrated an SVR12 (sustained virologic response at 12 weeks) of 96%, offering compelling evidence that co-infected persons can be treated for HCV with a once-daily regimen.
ION-4 is a Phase 3 study of co-infected patients with HCV genotype (GT) 1 and 4. The study was open to both HCV treatment-naïve and -experienced patients, and 20% of participants had compensated cirrhosis (early liver disease). Patients were on a variety of HIV regimens, and all had HIV RNA of less than 50 copies and CD4 cell count greater than 100.
Of the 335 patients, 276 (82%) were male, 115 (34%) were African American, 56 (17%) were Latino/a, and the average age was 52. The vast majority, 327 (98%), were GT1, and 185 (55%) were HCV treatment-experienced.
The study results showed high SVR rates, with no impact on HIV disease severity or treatment and minimal side effects. Overall, 321 of 335 patients, or 96%, achieved an SVR12. A breakdown of the SVR rates by treatment experience and sustained virologic responses in ION-4 are as follows:
ION-4
SUSTAINED VIROLOGIC RESPONSES
Overall SVR: 96%
Treatment-naïve 95%
Treatment-experienced: 97%
No cirrhosis: 96%
Cirrhosis: 94%
There were 10 HCV viral relapses and two treatment failures (both had poor adherence to the regimen), while one person was lost to follow-up and another died of non-treatment related causes (injection drug-related fatality). The 10 patients who relapsed were all African American. There were no clear explanations as to why their virus came back. Previous research has shown high response rates with this regimen in African Americans who were HCV mono-infected. This effect will be monitored going forward to see if this regimen does not perform as well in HIV/HCV co-infected African Americans as it does in other groups.
All patients maintained stable CD4 counts both during treatment and afterwards, and no patient experienced an HIV virologic rebound. The regimen was very well tolerated, with 257 (77%) reporting some side effects, but all were on the mild to tolerable level. Reported side effects included headaches, fatigue, diarrhea, nausea, arthralgia (joint pain), and upper respiratory tract infections. No one discontinued HCV treatment due to these side effects.
The results of the ION-4 study are very promising for patients with HIV/HCV co-infection and show that a once-daily regimen of LDV/SOF can cure people at very high rates with minimal side effects, and no impact on their HIV care. Although LDV/SOF has not been FDA approved for HIV/HCV co-infection, these results will further support patients and providers who wish to treat HCV off-label.
The ALLY-2 study, a Phase 3 clinical trial evaluating the effectiveness of daclatasvir (DCV) and sofosbuvir (SOF, brand name Sovaldi) for 8 or 12 weeks for patients living with HIV/HCV co-infection, shows very promising results for people with genotypes 1–4 with 12 weeks of therapy.
Daclatasvir is a pangenotypic (it works against GT 1–4) NS5A inhibitor taken once per day that is under review in the United States. Sofosbuvir, also pangenotypic, is an NS5B inhibitor that is taken once daily that has been FDA approved since late 2013. Both drugs are safe and well tolerated, and both have few drug-drug interactions, making them ideally suited for use in people living with HIV.
ALLY-2 comprised 203 patients with HIV/HCV co-infection, with genotypes 1–6. Study participants were given DCV plus SOF for 8 or 12 weeks. There were 151 treatment-naïve patients who were randomized into the 8-week arm (50 participants) or 12-week arm (101 participants). There were 52 treatment-experienced patients (all of whom took an interferon-based regimen in the past) who were all given 12 weeks of treatment. Most (98%) were on HIV ARTs, with HIV under control and high CD4 cell counts.
The 8-week results were not promising: 76% of patients achieved an SVR. The patients who took 12 weeks of DCV and SOF, however, achieved an overall SVR12 of 97%. A breakdown by genotype is in the chart below:



