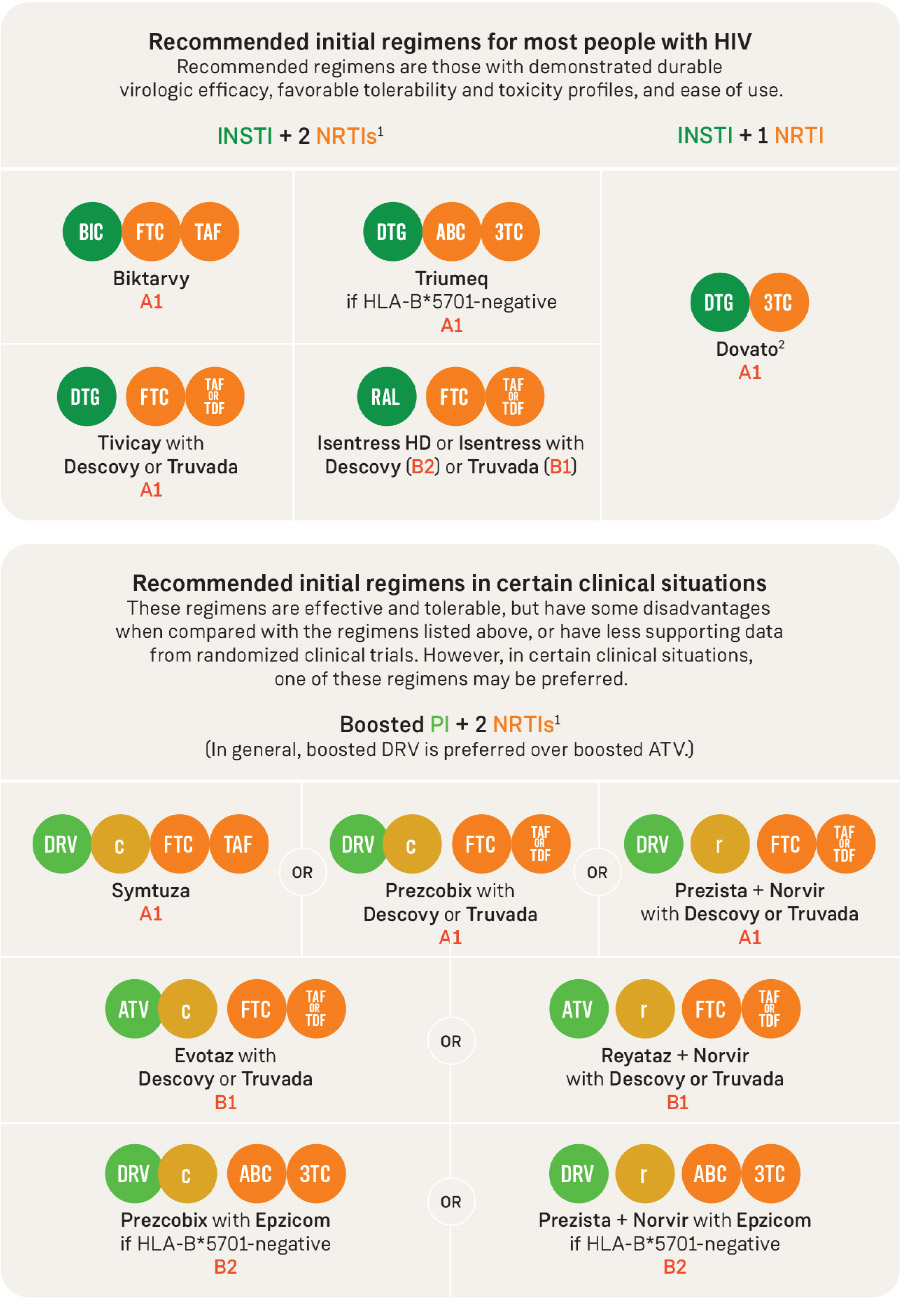
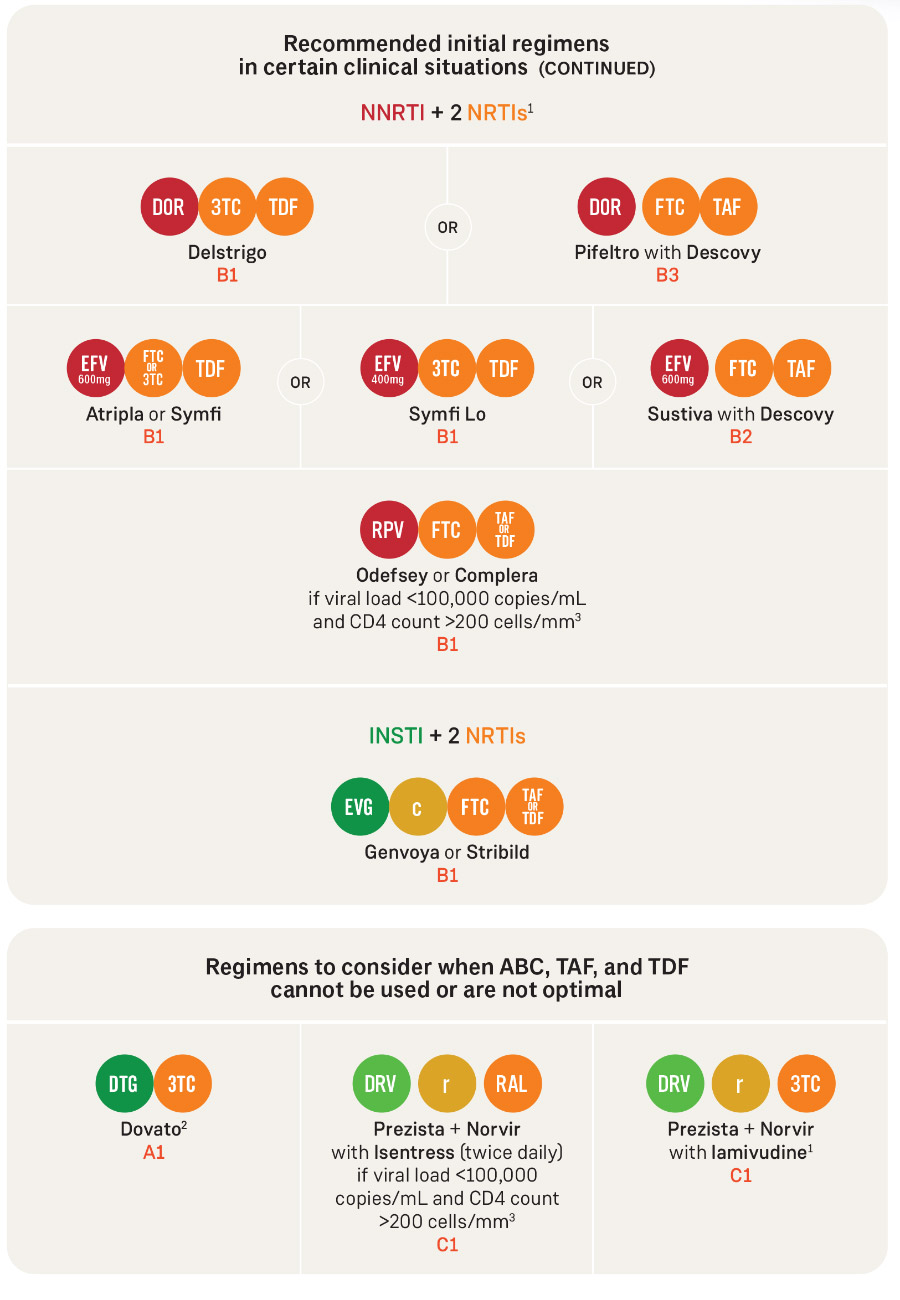
DHHS recommends starting antiretroviral therapy (ART) as soon as possible after HIV is diagnosed, regardless of CD4 count. Most people starting HIV treatment for the first time (treatment-naïve) should take one of the following: Biktarvy; Dovato; Triumeq; Tivicay plus Descovy or Truvada; or Isentress HD or Isentress plus Descovy or Truvada. Go to aidsinfo.nih.gov for more information.
Rating of recommendations
A: Strong
B: Moderate
C: Optional
Rating of evidence
1: Data from randomized controlled trials.
2: Data from well-designed non-randomized trials, observational cohort studies with long-term clinical outcomes, relative bioavailability/bioequivalence studies, or regimen comparisons from randomized switch studies.
3: Expert opinion.
Key to acronyms
3TC: lamivudine
ABC: abacavir
ART: antiretroviral therapy
ARV: antiretroviral
ATV: atazanavir
ATV/c: atazanavir/cobicistat
ATV/r: atazanavir/ritonavir
BIC: bictegravir
CD4: CD4 T lymphocyte, “T cell”
DOR: doravirine
DRV: darunavir
DRV/c: darunavir/cobicistat
DRV/r: darunavir/ritonavir
DTG: dolutegravir
EFV: efavirenz
EVG: elvitegravir
EVG/c: elvitegravir/cobicistat
FDA: Food and Drug Administration
FTC: emtricitabine
HLA: human leukocyte antigen
INSTI: integrase strand transfer inhibitor
NNRTI: non-nucleoside reverse transcriptase inhibitor
NRTI: nucleoside reverse transcriptase inhibitor
PI: protease inhibitor
RAL: raltegravir
RPV: rilpivirine
STR: single-tablet regimen
TAF: tenofovir alafenamide
TDF: tenofovir disoproxil fumarate
Note: The following are available as co-formulated drugs (Not a complete list)
Cimduo or Temixys: 3TC/TDF
Epzicom: ABC/3TC
Evotaz: ATV/c
Biktarvy: BIC/FTC/TAF
Delstrigo: DOR/3TC/TDF
Prezcobix: DRV/c
Symtuza: DRV/c/FTC/TAF
Dovato: DTG/3TC
Triumeq: DTG/ABC/3TC
Symfi Lo: EFV 400 mg/3TC/TDF
Symfi: EFV 600 mg/3TC/TDF
Atripla: EFV/FTC/TDF
Genvoya: EVG/c/FTC/TAF
Stribild: EVG/c/FTC/TDF
Descovy: FTC/TAF
Truvada: FTC/TDF
Odefsey: RPV/FTC/TAF
Complera: RPV/FTC/TDF
Footnotes
1. Lamivudine (3TC) may substitute for emtricitabine (FTC) or vice versa.
2. Except for individuals with pre-treatment HIV RNA greater than 500,000 copies/mL; who are known to have active hepatitis B virus (HBV) coinfection; or who will initiate ART before results of HIV genotype testing for reverse transcriptase or HBV testing are available.
Notes:
Tenofovir alafenamide (TAF) and tenofovir disoproxil fumarate (TDF) are two forms of tenofovir that are approved by the Food and Drug Administration. TAF has fewer bone and kidney toxicities than TDF, while TDF is associated with lower lipid levels. Safety, cost, and access are among the factors to consider when choosing between the two.
raltegravir (RAL) can be given as RAL 400 mg twice daily or RAL 1,200 mg (two 600 mg tablets) once daily.